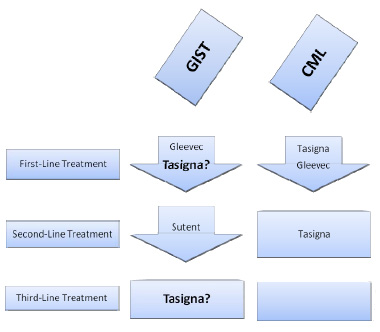
New target found for wild-type and pediatric GIST
A new target has been found for wild-type and pediatric GIST. GISTs without mutations in either of the two genes commonly mutated in GIST typically respond poorly to Gleevec. Andrew Godwin, Ph.D. of Fox Chase Cancer [...]