Biology
People with familial GIST inherit a mutated gene from one of their parents. It is a very rare type of GIST that affects up to two dozen families in the world (this number is subject to change as we learn more about GIST). Unlike sporadic GIST, people with familial GIST have KIT or (in one case) PDGFRA mutations in every cell in their body.
-
Chromosomes from a human male
There is another type of familial GIST called Carney-Stratakis Syndrome. These patients also can get another type of cancer called paragangliomas. This type of familial GIST is caused by mutations in one of the subunits of the succinate dehydrogenase (SDH) protein. For more information see Carney-Stratakis Syndrome.
The rest of this page will only discuss familial GISTs with KIT mutations; most of the behavior patterns of familial GIST with PDGFRA mutations are similar. In the case of KIT, different diseases, such as GIST, piebaldism or mastocytosis can occur depending on where the mutation occurs in the KIT gene.
Humans have two copies of every chromosome except the X and Y chromosomes. They also have two copies of the KIT gene, one on each copy of chromosome 4.
Children inherit one copy of each gene from each parent. This means that a child of someone that has familial GIST will almost certainly inherit a normal copy of the KIT gene from the parent that does not have familial GIST (since both copies of the gene are normal) and has a 50% chance of inheriting the mutant KIT gene from the parent that does have familial GIST. Individuals affected with familial GIST typically have one normal KIT gene (called wild-type) and one mutant KIT gene.
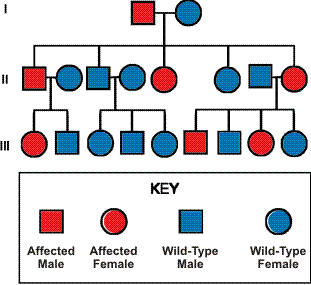
Inheritance pattern of familial GIST (autosomal dominant) (Adapted from Wikipedia)
Genetics
Familial GIST seems to be passed on in an autosomal dominant manner and has a high penetrance2. This means that children that inherit the mutant KIT protein have a high chance of developing GIST sometime during their lives. There has been at least one case however, in which an affected individual did not develop GIST until late in life. In this case, the proband (a woman) was diagnosed with GIST at age 37, her mother was diagnosed at age 57, and her grandmother was diagnosed at age 80. This family had a KIT exon 11 mutation that resulted in the deletion of codon 579. In another family, “the probability of being clinically diagnosed with GIST was 91% by age 70 among documented obligate carriers of the family mutation.”
Sporadic GIST
The vast majority of patients with GIST did not inherit mutations that cause GIST from their parents. Instead the initiating mutation, typically in the KIT gene but sometimes in the PDGFRA gene, developed in one or more cells sometime during their life. This mutation started a chain of events that eventually developed into GIST. Importantly, the KIT or PDGFRA mutations occur only in the tumor cells. The rest of the cells in the body have KIT and PDGFRA genes, but they are not mutated.
Diagnosis
Like sporadic GIST, familial GIST has often been misdiagnosed in the past. With the discovery of the role of KIT in 1998 and the approval of Gleevec for GIST in 2002, it is much more likely that a correct diagnosis will be made. Historically, familial GIST may have been classified as leiomyosarcoma, neurofibromatosis, “sarcomatosis”, leiomyomas, and possibly others.
The initial symptoms may include: anemia, fatigue, abdominal fullness or bloating, and difficulty swallowing (dysphagia). The skin may also be affected in several ways including hyperpigmentation (dark areas). The hyperpigmentation has typically been seen on the digits, elbows, knees, face and groin. In general, the pigmentation has first become evident in childhood or puberty, with a tendency to fade somewhat with aging.
Further testing such as endoscopy, ultrasounds and CT scans can help identify abnormalities. Tissue samples from abnormal tissue or masses are usually collected in a process known as a biopsy. Biopsies are used to aid in the diagnosis of cancer and GIST. Tissue samples are prepared from the biopsy sample.
One method that the pathologist uses to classify tissues is called immunohistochemistry. Using this technique, the pathologist applies various “antibodies” to the tissues. These antibodies are each designed to react with specific features (proteins) on the cell surface. The most important antibody that is applied when GIST is suspected is the KIT antibody. When these antibodies bind to their specific target on the cell surface they produce a “stain” or change in color of the sample. So when the KIT antibody is applied, if the cell surface has the KIT protein present on the surface, the sample will “stain positive”. This is what is known as “KIT positive” and means that this cell has KIT receptors on its surface. With very rare exceptions, GIST tumors will stain positive for KIT. This means that the cell is manufacturing or using the KIT protein.
Once a diagnosis of GIST has been obtained for the initial patient, other testing procedures might be used to identify affected individuals if familial GIST is suspected (see genetic counseling and mutational testing).
Natural History
Since KIT mutations are found in every cell of the body in familial GIST, the disease tends to have a different natural course than sporadic GIST. In sporadic GIST, a tumor will tend to start in one location in the GI tract. If found and removed early enough the patient may be cured. For many patients however, the GIST will spread to other sites through a process known as metastasis. This probably occurs as cells detach and spread from the original tumor into the blood and are distributed to other sites in the body where they find a new home and eventually grow new tumors.
In familial GIST, the cells along the GI tract may undergo changes at multiple locations since all of the cells have the KIT mutations. Since the KIT receptor/protein is not important to many cell types, only certain types of cells, such as the interstitial cells of cajal (ICC), will be negatively affected. ICC are the pacemaker cells of the GI tract and are responsible for providing the stimuli that cause the muscles of the GI tract to contract and move food through the GI tract. ICC express the KIT protein and is known to be important to their function and life cycle. As a result of all of the ICC cells having KIT mutations, tumors tend to form in more than one location in the GI tract as opposed to one primary tumor in the GI tract (with possible metastases outside the GI tract). Prior to the formation of tumors, the affected areas/cells may take on a pre-cancerous appearance known as “hyperplasia”.
Despite the tendency for multiple tumors to develop in the GI tract, familial GIST may have less of a tendency to spread via metastasis. The clinical course has been described as “relatively indolent”. In spite of this description of one large family by Robson et al., another description by Kleinbaum et al. of a family with 15 suspected cases, described a “metastatic phenotype”.
In one large family (Robson et al.)with 22 presumed carriers, “metastatic disease has rarely been a prominent feature of the clinical course” and “a number of individuals have survived to later ages. . . “. The paper referenced also noted, “in at least 2 cases, index lesions appear to have grown minimally over periods of as long as 10 years.” In this large family, “the diagnosis of GIST appears to have been made at a progressively younger age in successive generations of this family. The mean age at diagnosis in the most recent generation has been 34 years, compared with 47 years in the middle generation and 62 years in the oldest. Nonetheless, the predisposition cannot be considered innocuous. Several individuals have required prolonged hospitalizations as a result of gastrointestinal bleeding, and 3 family members are believed to have died from obstructive complications of their disease.”
Treatment
GIST is a rare disease. It is recommended to seek consultation at a center that has extensive GIST expertise. This is even more important with familial GIST as it is even rarer and does not have the same natural course as sporadic GIST.
Robson et al. note in their series “Whereas careful monitoring for the development of GIST is reasonable, the indolent clinical course and multifocality of the disease suggests that surgical intervention should be deferred in the absence of complications. The role of imatinib (Gleevec) as a therapeutic or preventive intervention in these rare individuals remains to be defined.”
We are aware of several anecdotal cases of familial GIST patients taking Gleevec. Kleinbaum et al. describe familial GIST in their “family” as having “in vivo sensitivity to imatinib therapy.”
Genetic Counseling
Because of the many factors to consider about familial GIST, individuals may want to seek genetic counseling. This would typically be recommended soon after familial GIST is suspected.
Genetic counseling is the process by which patients or relatives at risk of an inherited disorder are advised of the consequences and nature of the disorder, the probability of developing or transmitting it, and the options open to them in management and family planning in order to prevent, avoid, or ameliorate it. This complex process can be seen from diagnostic (the actual estimation of risk) and supportive aspects.
Genetic counselors work as members of a health care team, providing information and support to families who have members with birth defects or genetic disorders and to families who may be at risk for a variety of inherited conditions. They identify families at risk, investigate the problems present in the family, interpret information about the disorder, analyze inheritance patterns and risks of recurrence and review available options with the family. Patients seeking genetic counseling are encouraged to contact Project Flag (see top of page).
Mutational Testing
At some point, usually after appropriate genetic counseling, family members might be offered the option of genetic testing. If the proband has had mutational testing (a common test for sporadic GIST), then they will know what type of KIT or PDGFRA mutation they have. It is then possible to test for this same mutation in family members. DNA is needed from the person undergoing testing. This can be taken from a mouth swab in some cases or from blood. The DNA is then screened to see if it contains the same mutation as the proband.
Links for Familial GIST
Novel c-KIT germline mutation in a family with gastrointestinal stromal tumors and cutaneous hyperpigmentation
Miguel Carballo, Ignasi Roig, Francesc Aguilar, Maria Antonia Pol, María José Gamundi, Imma Hernan, María Martinez-Gimeno
- This is the 2004 report of a Spanish family (n=?) with a germline mutation in KIT exon 11; a duplication which produced two extra glutamine and leucine residues between positions 576 and 577.
Gastrointestinal stromal tumors in a mouse model by targeted mutation of the Kit receptor tyrosine kinase (full text)
Gunhild Sommer, Valter Agosti, Imke Ehlers, Ferdinand Rossi, Selim Corbacioglu, Judith Farkas, Malcolm Moore, Katia Manova , Cristina R. Antonescu, and Peter Besmer
- This mouse model is essentially a model of familial GIST. The mice pass on the KIT mutations to their offspring.
PDGFRA germline mutation in a family with multiple cases of gastrointestinal stromal tumor
Agnès Chompret, Caroline Kannengiesser, Michel Barrois, Philippe Terrier, Philippe Dahan, Thomas Tursz, Gilbert M. Lenoir and Brigitte Bressac-De Paillerets
- This (France) abstract describes a kindred of 5 patients with GIST. A germ-line PDGFRA missense mutation, 2675G > T, resulting in a tyrosine substitution for the highly conserved aspartic acid at codon 846. Interestingly, PDGFRA Asp846 is homologous to codon 820, which is located in the KIT tyrosine kinase II domain. In a previous study, a KIT germline Asp820Tyr mutation was detected in a Japanese kindred in which 6 individuals had GIST.
Intestinal neurofibromatosis is a subtype of familial GIST and results from a dominant activating mutation in PDGFRA
de Raedt T, Cools J, Debiec-Rychter M, Brems H, Mentens N, Sciot R, Himpens J, de Wever I, Schoffski P, Marynen P, Legius E.
- This 2006 study found an inherited mutation (Y555C) in the juxtamembrane domain of PDGFRA in the affected individuals. The Y555C mutation leads to autophosphorylation and thus activation of PDGFRalpha. These observations confirm that PDGFRalpha(Y555C) is an oncogenic kinase. The clinical phenotype in the reported family resembles the syndrome of familial gastrointestinal stromal tumors (familial GIST).
Familial gastrointestinal stromal tumors associated with dysphagia and novel type germline mutation of KIT gene
Seiichi Hirota, Toshirou Nishida, Koji Isozaki, Masahiko Taniguchi, Kazuhiro Nishikawa, Akiko Ohashi, Arimichi Takabayashi∥, Tadashi Obayashi, Tomoko Okuno, Kazuo Kinoshita, Hui Chen, Yasuhisa Shinomura and Yukihiko Kitamura
- This 2002 paper describes a family with germline mutation of KIT (Asp-820 in TKII). Affected family members had dysphagia (trouble swallowing). They had a thickened hyperechoic layer between the circular and longitudinal muscle layers, suggesting hyperplasia of ICC at the myenteric plexus layer.
Gene Expression in Gastrointestinal Stromal Tumors Is Distinguished by KIT Genotype and Anatomic Site
Cristina R. Antonescu, Agnes Viale, Lisa Sarran, Sylvia J. Tschernyavsky, Mithat Gonen, Neil H. Segal, Robert G. Maki, Nicholas D. Socci, Ronald P. DeMatteo and Peter Besmer
- This 2004 report lists one familial case with a KIT exon 11 W557R mutation. It also discusses some of the differences in gene expression between sporadic GISTs and familial GISTs.
References
A novel K509I mutation of KIT identified in familial mastocytosis-in vitro and in vivo responsiveness to imatinib therapy, Zhang LY, Smith ML, Schultheis B, Fitzgibbon J, Lister TA, Melo JV, Cross NC, Cavenagh JD.
Analysis of KIT Mutations in Sporadic and Familial Gastrointestinal Stromal Tumors:Therapeutic Implications through Protein Modeling (Full text PDF file), Chi Tarn,1ErinMerkel,1Adrian A. Canutescu,2 Wei Shen,1Yuliya Skorobogatko,1MartinJ. Heslin,4 Burton Eisenberg,5 Ruth Birbe,3 Arthur Patchefsky,3 Roland Dunbrack,2 J. PabloArnoletti,4 Margaret vonMehren,1andAndrewK. Godwin1
- In this 2005 article, a family (n=4) with a germ-line KIT exon mutation with deletion of amino acid 579 is described.
Pleomorphic Characteristics of a Germ-Line KIT Mutation in a Large Kindred with Gastrointestinal Stromal Tumors, Hyperpigmentation, and Dysphagia (full text), Mark E. Robson, Emily Glogowski, Gunhild Sommer, Cristina R. Antonescu, Khedoudja Nafa, Robert G. Maki, Nathan Ellis, Peter Besmer, Murray Brennan, and Kenneth Offit
- This 2004 paper describes a large family of Italian descent (n=22 presumed carriers) with germ-line mutation of W557R (KIT exon 11).
Familial gastrointestinal stromal tumor with homo-/hemizygous kit exon 11 deletion: Genotypic, histopathologic, radiographic, and therapeutic findings, E. P. Kleinbaum, L. L. Chen, A. Lazar, L. Strong, W. Zhang, H. Choi, R. Benjamin, S. Patel, J. C. Trent
- This ASCO abstract describes a large U.S. family (n=15) with a germ-line KIT exon mutation with deletion of amino acid 579.
