The time it takes between the appearance of first symptoms of a rare disease and when the correct diagnosis is given, is often years if not decades instead of days or weeks like it is for a more common illness.
When that rare disease is identified, like in the case of GIST, very straightforward questions are asked, such as: Who gets GIST? Is it the same treatment for every patient? Will the cancer return after initial treatment? How long should a patient stay on a particular drug? Can I get off the treatment since I’ve had no evidence of recurrence?
There was not sufficient information to accurately answer those questions years ago and some questions still remain unanswered today. This is where the LRG Patient Registry is so powerful, particularly for GIST. The natural history of the disease has been observed over time by collecting patient reported ongoing and supporting medical information on well-defined outcomes of interest for analysis and reporting. It has become an efficient tool for clinicians, patients and caregivers in understanding the long term turn out – either with or without treatment(s).
The Empowered Patient
Patient groups such as the LRG can design and conduct their own studies, collect their own data, analyze the results, and publish the research. They can provide researchers an access to targeted study populations at little or no cost.
Since its creation, the LRG Patient Registry has facilitated the gathering together of patients and their families or caregivers all over the world as they engage directly and indirectly into the development of the very database in which their data will be entered. It serves as a key instrument for building and empowering the GIST patient community.
Patients can contribute to improving the robustness, comprehensiveness and quality of the patient registry by reporting their surveillance and treatment data along with evaluation reports provided by their healthcare professionals. The collected data has empowered patients with inferred information based on analysis and population experience in making crucial treatment decisions together with their healthcare providers.
With active patient participation and a well-implemented registry, the likelihood for developing a cure for GIST is increased. Furthermore, the consistent collection of patient data leads to better standards of care which will dramatically improve patient outcomes and life expectancy even in the absence of new therapies.
Importance of Information
Biological samples have become valuable due to well-documented, associated data housed in the patient registry.
The Patient Registry is linked with the GIST Collaborative Tissue Bank through an online system that is maintained in a secure server. A unique identifier is assigned to each patient to facilitate data linkage and provides authorized access to researchers to an anonymized dataset. Soon, patients will be able to access their own records and update them.
The information in the Patient Registry has been an indispensable infrastructure tool for translating basic and clinical research into the improvement of patient care and treatment planning and quality of life outcomes in GIST.
A Glimpse of the Patient Registry
Information on patients’ specific mutation, symptoms, previous treatments, ongoing evaluations and treatments pertaining to GIST, ethnicity, age and tumor type at diagnosis are just a few of the types of data that is being reported in the Patient Registry. By recording such a wide range of information, the registry provide insight on the epidemiology or any genotype correlations; whether certain treatments have better outcomes; risk assessment and prognosis, survival and many others. Through this patient- driven database, it allows researchers to more accurately estimate and to determine the natural history, providing answers to many questions. Once this information is known, it can be used to give more accurate advice to patients, and optimize care pathways.
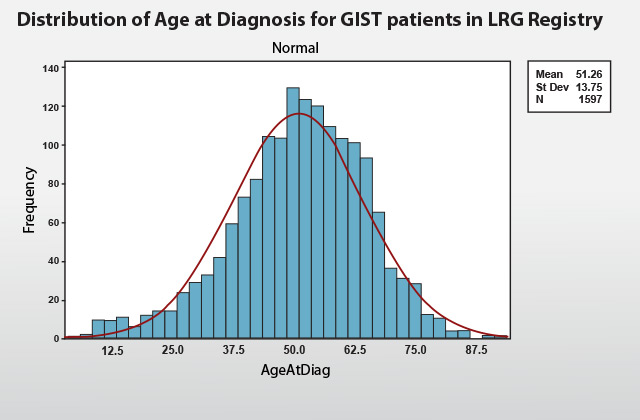
In the LRG Patient Registry, 65 percent of the group at which GIST with random mutation is found in the age between 36 to 64 years old. Children are affected very rarely and the earliest age is 5.5 years old. Persons with inactivation of the neurofibromatosis 1 gene (NF1) are more likely to develop GIST than the general population and are more likely to be diagnosed at younger ages (however, NF1-associated GIST is very rare in the LRG registry). Familial GIST also develops in younger or middle-aged adults. The latest age that GIST was diagnosed in the group is at 92 years old.
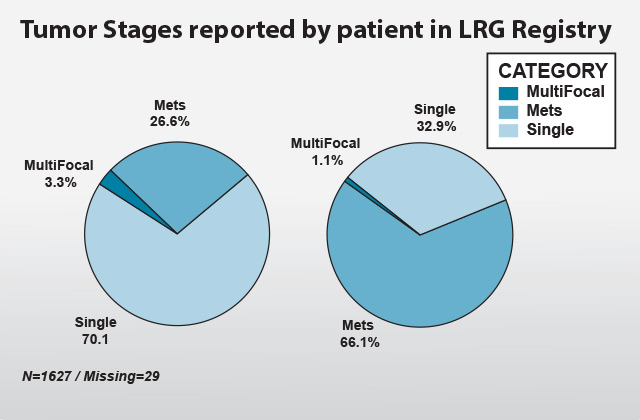
Identifying the stage of tumor at diagnosis is important in selecting the appropriate treatments as well as in estimating prognosis. Staging describes the severity of a person’s cancer based on the size and/or extent (reach) of the original (primary) tumor and whether or not cancer has spread in the body. In the LRG Patient Registry, 70.1 percent of patients reported single tumor at diagnosis. As patients were monitored over time, 37.2 percent of the single tumors and 2.2 percent of multifocals at diagnosis group develop recurrence or metastasis later. Currently, total patients at metastatic stage are at 66.1 percent.The occurrence of multiple lesions in the same organ is called multifocal; it is uncommon and restricted to pediatric GISTs and rare hereditary conditions.
MutiFocal refers to a presentation state; it should not really change
Some tumors can be removed by surgery and pose little risk of coming back. Other tumors return after surgery, called a recurrence.
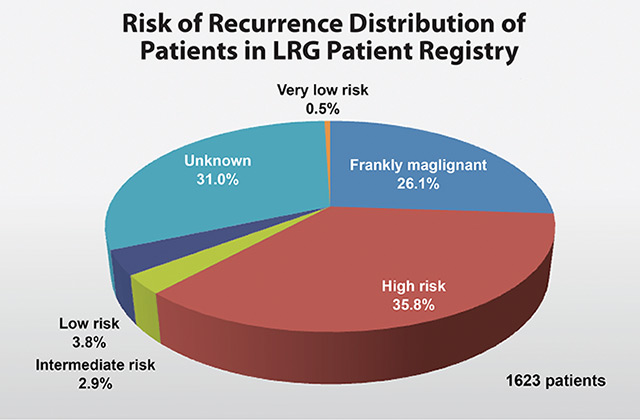
In the LRG Patient Registry, the risk of recurrence is classified using the Modified NIH Method which factors the primary tumor size and location, mitotic activity and rupture of the tumor either prior to or during surgery. Some patients may not know or have access to one or two of the parameters. Risk assessment can sometimes still be made based on available details. The chart above shows the patient distribution (n=1623) based on risk of recurrence at the time of presentation. The highest percentage (35.8 percent) represents the patients who have the highest risk of recurrence based on their reported diagnosis details. 26.1 percent represents those who are frankly malignant, and includes all those patients who were metastatic at diagnosis and are also considered high risk. Since the LRG patient registry highly relies on patient reported data, 31 percent are considered to have unknown risk. This may be due to the fact that some patients have been diagnosed early on and were not confirmed to have GIST till later, or their diagnosis details are no longer available or were not provided to the patient. Note: Pediatric-type GIST may have a very different potential for metastases.
Knowing your risk of recurrence is especially important if you are considering preventative Gleevec. Patients with a low risk or very low risk tumor may not need to take adjuvant Gleevec. At least three years of preventative Gleevec is recommended for GIST patients with a significant risk of recurrence (intermediate or high risk).
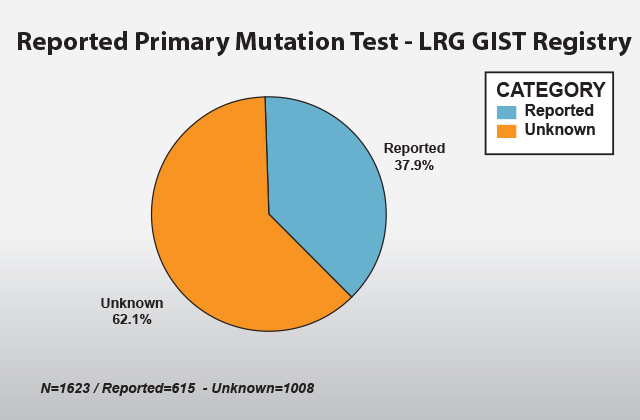
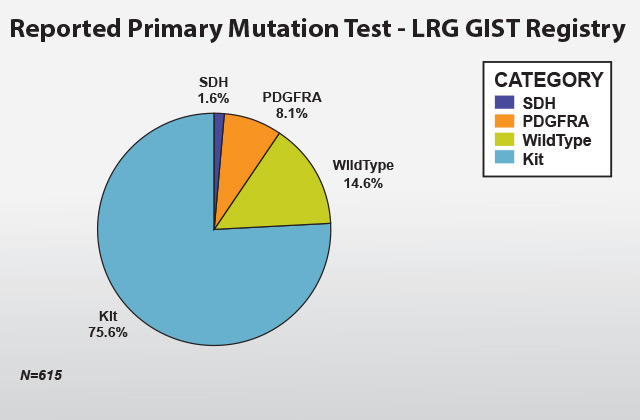
Based on those who reported mutation and those analyzed in the LRG tissue bank, 75.6 percent of the group have a mutation in the Kit gene. The most common mutation is in Exon 11, which is found in 81.3 percent of cases. Mutations in exon 11 generally respond to treatment with Gleevec better than mutations in other exons.
Exon 9 mutations are the second most common mutation found in about 15.3 percent of cases. In patients that have an exon 9 mutation, in most of the cases they occur in the small bowel or colon. GISTs with exon 9 mutations have a lower response rate to standard dose Gleevec therapy when compared to exon 11 mutations. As a result, a higher dose of Gleevec is generally recommended for patients with advanced/metastatic disease. They also seem to respond fairly well to Sutent.
Exon 13 and exon 17 mutations in the primary tumor are rare in the registry.
Some GIST tumor cells do not contain c-kit mutations, but a closely related gene, PDGFRA is mutated, and 8.1 percent of GIST cases in the patient registry have this type of mutation. About one-third of the PDGFRA mutations may still respond to Gleevec and/or Sutent, but up to two-thirds of PDGFRA mutations do not respond to these drugs. These mutations occur in one specific spot in exon 18 of the gene and are called a D842V mutation (PDGFRA mutation, exon 18, D842V).
GIST tumors that do not have a mutation in KIT or PDGFRA are called “wild-type” GIST. 14.6 percent of patients in the registry reported having this type. It was recently found that some of these tumors have mutations in other genes such as BRAF or SDH GIST (1.6.percent). Wild-type tumors do not respond as well as other types to Gleevec as it is not a very potent inhibitor of wild-type KIT and KIT may be less important. Some signaling may still occur in KIT and Gleevec treatment may be beneficial in some cases. More potent inhibitors of wild-type KIT, especially Sutent and Tasigna (nilotinib), may provide more benefit for wild-type GIST.
Despite the increasing importance of mutational testing over the years, 62.1percent of GIST patients in the registry have unknown mutations, some unreported while others did not have mutational testing performed on their tissue samples. Mutational testing is now recommended for metastatic and high-risk GIST patients
Data from Patient Registries such as the LRG’s has the potential to provide analytics that will generate the knowledge necessary to meet the needs of patients, physicians, clinicians and researchers, and eventually to find cures for diseases such as GIST.