A new gene panel takes mutation testing to a new level. The panel tests for mutations in 23 genes at once will help to properly classify wild-type GIST patients. 
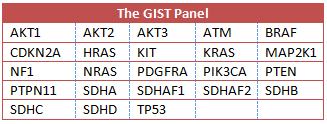
Working with next generation sequencing and in cooperation with Ion Torrent™ the new GIST panel is being developed by Christopher Corless, M.D., Ph.D., chief medical officer of the Knight Diagnostic Laboratories at Knight Cancer Institute, Oregon Health and Sciences University (OHSU). GIST patients can now be tested for mutations in 23 genes with one test (see Table 1). The Heinrich/Corless labs at OHSU have been leaders in genetic testing (genotyping) in the targeted treatment era (the Gleevec/Sutent/TKI era).
Wild-type GIST has been a diagnosis of exclusion. The current definition is a GIST tumor that does not have a KIT or PDGFRA mutation, the most common mutations in GIST. This definition may not be keeping up with research, however, as scientists have slowly been uncovering the underlying mutations associated with wildtype GIST. To date, mutations in wildtype GIST have been found in all four subunits of succinate dehydrogenase (SDH); SDHA, SDHB, SDHC and SDHD, as well as in BRAF, KRAS, HRAS and NRAS. Between 6 percent and 25 percent of patients with NF1 (or neurofibromatosis-related protein) may also develop GISTs. BRAF mutations might represent the most compelling reason for testing as BRAF inhibitors, used in melanoma, are clinically available. While the treatment path for those with SDH mutations is not as clear, establishing the correct diagnosis opens the possibility to try alternate therapies that are more directed at SDHrelated pathways, such as VEGF receptors and IGF1R.
While the new test won’t replace the current testing for KIT or PDGFRA mutations, it should greatly help in identifying a wide range of mutations that might occur in wild-type GIST. In addition to mutations that have already been published, the new panel will examine a number of other genes of interest. The new GIST panel will be available starting in August (2012).
 In some cases, a biopsy does not retrieve enough tumor tissue for mutation testing. The GIST panel uses paraffinbased tissue and requires less tissue than previous testing methods. It’s possible that some tissue that previously could not be analyzed due to insufficient amounts of tissue might contain enough tissue to be analyzed with the new test.
In some cases, a biopsy does not retrieve enough tumor tissue for mutation testing. The GIST panel uses paraffinbased tissue and requires less tissue than previous testing methods. It’s possible that some tissue that previously could not be analyzed due to insufficient amounts of tissue might contain enough tissue to be analyzed with the new test.
Immunohistochemistry (IHC), a technique long used to identify which proteins are present in tumor cells, may also have an updated role in identifying pediatric- type GIST as new stains for SDH are developed. However, the ability to stain for SDH may be limited to specialty centers.
For more information about this testing check out this article.



