Trial investigators for the Global Phase 3 GRID trial reported that the Bayer Pharmaceuticals’ drug regorafenib (BAY 73-4506) has the potential to fill the unmet need for advanced GIST patients progressing after imatinib and sunitinib.
Dr. George Demetri of the Dana Farber Cancer Institute was the leading Principal Investigator and presented the data for the team late in the afternoon on June 4. According to Dr. Demetri, “(regorafenib) potentially represents a new standard of care for this patient population.”
From January to July 2011, 236 patients were screened and 199 entered the GRID trial in 17 countries. Dr. Demetri attributed the rapid seven-month accrual to the high degree of international collaboration. We would add that GIST patients are typically knowledgeable about clinical trials and also willing to participate. In the trial patient profile 40- 45 percent of subjects had more than imatinib and sunitinib, and 21-30 percent had received nilotinib.
Progression Free Survival (PFS) was 4.8 Months (95 percent CI 4.1-5.8) and exceeded the trial design estimate targeting a 100 percent improvement. Median OS was not reached and because of cross-over design will probably not be significant. PFS benefit was significantly present for all sub-groups analyzed except for patients exposed to imatinib for less than six months where the benefit was not as clear.
To give some perspective on the reported results, the sunitinib Phase 3 trial reported PFS was 5.6 months. PFS appeared to be equal in both Exon 11 and Exon 9 sub-groups. This is similar to the limited data we have for sorafenib and in contrast to imatinib where Exon 11 seems to do better than Exon 9 at 400 mg and in contrast to sunitinib where Exon 9 seems to do better than exon 11.
We don’t have comparable data for sorafenib. However, very limited data from the Phase 2 trial (Kindler et. al. ASCO 2011) showed comparable overall beneficial response between exon 11 (n=22) and Exon 9 (n=4).
This data again raise the question of whether there is a better second line choice for Exon 11 than sunitinib…a question that can only be answered by a future head to head clinical trial. Note: this type of trial is rarely accomplished due to the number of subjects required, and the related cost.
The two Bayer drugs, regorafenib and sorafenib, are very similar in design (regorafenib differs by the addition of one fluorine atom) but behave differently as drugs. Regorafenib is taken in 160 mg per day doses for three weeks of a four week cycle versus sorafenib which is taken in twice daily in doses of 400mg each with no break. Regorafenib is also a relatively stronger inhibitor of c-KIT (~10X). Both drugs inhibit BRAF, c- KIT, PDGFRb, Raf, and VEGFR 2/3. Regorafenib differentially inhibits RET and VEGFR1, and to a lesser extent TIE2. Sorafenib differentially inhibits Flt3. Dr. Demetri reported that regorafenib has active metabolites that carry the drug’s effect into a one-week off period.
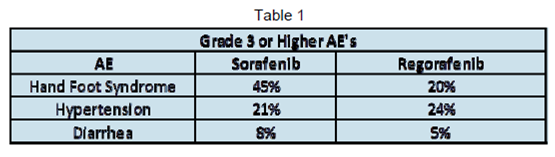
These differences may not have carried into the side effect profile. (See Table 1). Investigators reported “no new or unexpected safety findings with regorafenib”. Most significant Adverse Events (AEs) (Grade 3 and higher) on regorafenib were very similar to those reported at ASCO 2011 for the Phase 2 sorafenib trial.
Interestingly, Dr. Demetri reported that AE related drug discontinuation was proportionally higher in the Placebo group, where 6.1 percent of patients on regorafenib discontinued because of AE’s at median 23 weeks exposure and 7.6 percent of patients on placebo discontinued because of AE’s at median seven weeks exposure.
If regorafenib for GIST is approved by the FDA it will be the third drug approved for advanced GIST and the first for third-line use. It has been over six years since the last drug (sunitinib) was approved for advanced GIST for secondline use.
Two Other Drugs Clinical trial designs, and particularly controls, have figured into trials of two other drugs for GIST that have attempted to fill the third-line slot. IPI-504 Phase 3 in advanced GIST was terminated in April 2009, when the trial was stopped by the safety committee due to toxicity. The IPI-504 trial had a placebo control which arguably limited accrual and therefore patient exposure. At the June 2010 ASCO investigators reported that nilotinib Phase 3 in advanced GIST did not achieve its efficacy goals when other TKI’s were used as controls. The trial accrual process resulted in some nonprogressing patients entering the trial and affecting the outcome. But the residual efficacy of imatinib and/or sunitinib in the third line control group was a significant design hurdle for nilotinib to overcome.
*It is important to note that ASCO reports are considered conference reports and they are not the same as peer reviewed published reports. In the pantheon of evidence-based science, conference reports are somewhere below the gold standard–peer reviewed formally published data. They may eventually be published in a peer-reviewed journal, but until then they are not at the higher level of evidence.