This is the eighth article in a series written by each of the key members of the LRG Resistance Research Team.
Several articles in the LRG newsletter from our GIST Resistance Research Team have been written over the last year, focusing mainly on the role of KIT and PDGFRA mutation in the genesis and treatment of GIST. We know that KIT and likely PDGFRA are very important for the interstitial cells of Cajal (ICC), a cell that resides along the entire length of the intestinal tract and has the important function of coordinating peristalsis, the movement that propels food along the digestive tract. ICC strongly express KIT protein and loss of KIT results in loss of ICC. Much like the pacemaker cells of the heart, which coordinate contraction, ICC coordinate peristalsis through an inherent pacemaker function. After receiving a signal from the brain that food has been consumed, rhythmic contractions are set up with the help of the ICC that propel food along the gastrointestinal tract. Thus, the ICC serve a critical function in normal gastrointestinal function. As you might imagine, ICC are known or thought to be involved in many diseases of the gastrointestinal tract. There are many lines of evidence that have shown that an activating mutation in KIT or PDGFRA within ICC or a related pre-ICCtype cell, causes the cell to proliferate in an uncontrolled fashion, resulting in a GIST. KIT or PDGFRA mutation is said to be the initiating event (tumor initiation) that gives rise to GIST. We know that KIT or PDGFRA mutation is important because when we target them with a drug (eg. Gleevec/ Imatinib mesylate or Sutent/ Sunitinib maleate), GISTs stop growing. However, KIT or PDGFRA mutation is “the tip of the iceberg”in terms of important genetic changes that are found in GIST. Several more of these changes take place in GISTs after initiation by KIT or PDGFRA mutation. In aggregate these changes are involved in what is known generically as tumor progression.
While I don’t want to belittle tumor initiation – after all, understanding tumor initiation has led to some amazing therapeutic options for the treatment of GIST – tumor progression is what takes a rather innocuous tumor that is not very likely to do much harm to anyone and turns it into a nasty cancer capable of moving within the body (metastasis) to sites like the liver and in the end, is what enables a cancer to kill the patient. Therefore, it is likely that to truly cure GIST we will need to understand tumor progression. While much is known about tumor progression in other systems, very little is known in GIST.
Genes/proteins involved in tumor progression can be grouped into tumor suppressors and oncogenes. The normal function of tumor suppressors is to stop cells from dividing/proliferating inappropriately while the normal function of oncogenes is to facilitate cellular division/ proliferation, cell migration, etc.. Generally, inactivating mutations arise in tumor suppressors, resulting in increased/ inappropriate cell division/ proliferation while oncogenes generally have activating mutations and/or are amplified genetically so there is a lot more protein around than is normal.
TP53 (known as P53) is a well-known tumor suppressor involved in many types of cancer. Its job is to prevent cells from dividing inappropriately, as in the case of a cell with extensive DNA damage. RAS is a well known oncogene. RAS seems to control growth through positive actions. Much of what we know about tumor progression in GIST comes from cytogenetic/ chromosomal analysis of GISTs. Jonathan Fletcher and Maria Debiec-Rychter from the GIST Resistance Research Team are expert cytogeneticists and have both contributed many karyotypes/cytogenetic profiles of GISTs. Cytogeneticists examine the chromosome number and structure of cells to identify regions that are abnormal. Humans have twenty three pairs of chromosomes, twentytwo pairs of autosomes (numbered 1-22) and one pair of sex chromosomes (designated X or Y). Men have an X and a Y chromosome while women have two X chromosomes. Each chromosome has a short arm (designated “p) and a long arm (designated “q”). Other techniques have also been used to identify chromosomal regions that are consistently abnormal in GIST including Fluoresecence in-situ Hybridization, Loss of Heterozygosity Studies and Comparative Genomic Hybridization. A description of these techniques is beyond the scope of this article but essentially, they allow a more fine structure analysis of chromosomes than cytogenetic analysis. All of these studies are complementary.
KIT and PDGFRA both reside next to each other on the long arm of chromosome 4 but this region of chromosome 4 looks normal in GIST. This is 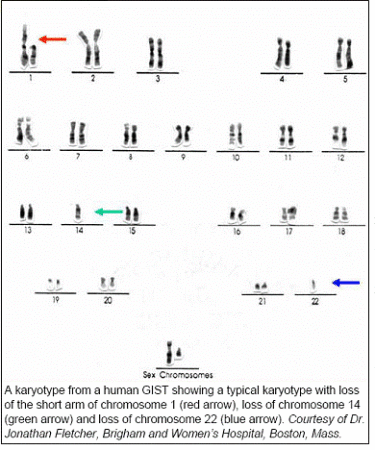 because GISTs are highly dependent on fulllength KIT or PDGFRA in most cases and the genetic changes we find in KIT and PDGFRA are too small to be seen by these types of studies, which require very high resolution studies such as DNA sequence analysis. However, other chromosomal changes are relatively common in GIST. Several chromosomes and chromosomal regions have been implicated in tumor progression. The most common regions of chromosomal loss are the long arms of chromosomes 14 and 22 and the short arms of chromosome 1 and 9 and the most common region of chromosomal gain is the long arm of chromosome 8 (see Figure 1). Interestingly, loss of material from the short arm of chromosome 9 is associated with aggressive/malignant behavior.
because GISTs are highly dependent on fulllength KIT or PDGFRA in most cases and the genetic changes we find in KIT and PDGFRA are too small to be seen by these types of studies, which require very high resolution studies such as DNA sequence analysis. However, other chromosomal changes are relatively common in GIST. Several chromosomes and chromosomal regions have been implicated in tumor progression. The most common regions of chromosomal loss are the long arms of chromosomes 14 and 22 and the short arms of chromosome 1 and 9 and the most common region of chromosomal gain is the long arm of chromosome 8 (see Figure 1). Interestingly, loss of material from the short arm of chromosome 9 is associated with aggressive/malignant behavior.
The area on chromosome 9 that is of particular interest has been narrowed down to two well-known genes known as CDKN2A and CDKN2B. These two genes encode three important cell regulatory proteins known as p16(INK4A), p14(ARF), and p15(INK4b). The accumulated evidence so far suggests that loss of both p16(INK4A) and p14(ARF), which are both encoded by the CDKN2A gene, is very important in tumor progression in GIST. p16(INK4A) and p14 (ARF) control important checkpoints in the cell division cycle. Checkpoints are points within the cell division cycle where a cell decides whether to divide or not. It is not hard to imagine that loss of proteins responsible for telling the cell to stop dividing would be advantageous for a cancer. If one were to think of a GIST as a car, I might suggest that KIT or PDGFRA activating mutations are the equivalent of pushing the gas pedal to the floor (pedal to the metal) while inactivation of CDKN2A is like removing the brakes. This results in a car (or tumor) that is accelerating out of control without the ability to stop. A major challenge is how we might exploit loss of CDKN2A therapeutically. As far as I know, therapeutics have not been designed that enable one to re-establish p16(INK4A) and/or p14(ARF) function. Of course, the other chromosomal regions with consistent losses and gains are being studied by several groups so hopefully, one or more of those areas will present a good opportunity for a targeted therapy that could be used independently or in conjunction with Gleevec or Sutent. Through this work and others, we are all hoping that some day soon we will be able to cure GISTs.
References
1.Rubin BP, Heinrich MC, Corless CL. Gastrointestinal stromal tumour. Lancet. 2007;36:91.
2. Perrone F, Tamborini E. Dagrada GP, Colombo F et al. 9p21 locus analysis in high-risk gastrointestinal stromal tumors characterized for c-kit and plateletderived growth factor receptor α gene alterations. Cancer 2005;104:159.



